Modifications on the basic skeletons of vinblastine and vincristine
- PMID: 22609781
- PMCID: PMC6268133
- DOI: 10.3390/molecules17055893
Modifications on the basic skeletons of vinblastine and vincristine
Abstract
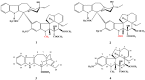
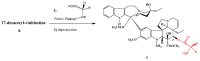
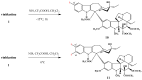
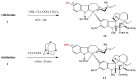
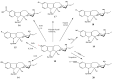
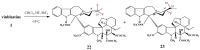
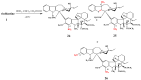
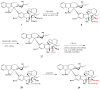
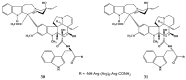
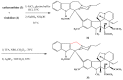
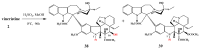
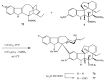
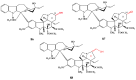
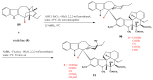
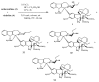
The synthetic investigation of biologically active natural compounds serves two main purposes: (i) the total synthesis of alkaloids and their analogues; (ii) modification of the structures for producing more selective, more effective, or less toxic derivatives. In the chemistry of dimeric Vinca alkaloids enormous efforts have been directed towards synthesizing new derivatives of the antitumor agents vinblastine and vincristine so as to obtain novel compounds with improved therapeutic properties.
Figures


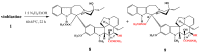

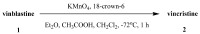
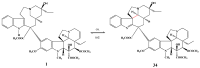
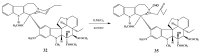
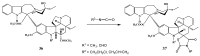
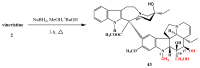

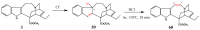
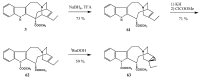
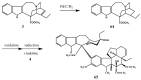
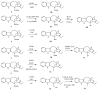

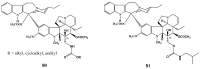

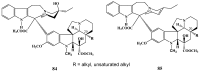
References
-
- Brossi A., Suffness M. The Alkaloids. Vol. 37. Academic Press Inc.; New York, NY, USA: 1990. Antitumor Bisindol Alkaloids from Catharanthus Roseus (L.) pp. 1–240.
-
- Bölcskei H., Szabó L., Szántay C. Synthesis of vinblastine derivatives. Front. Nat. Prod. Chem. 2005;1:43–49. doi: 10.2174/1574089054583849. - DOI
-
- Eli Lilly Company, Neue Aminderivate von Vinblastin, Leurosidin und Leurocristin und Verfahren zu ihrer Herstellung. 22415980. DE Patent. 1974 [Chem. Abstr. 1974, 82, 579967b]
-
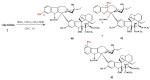
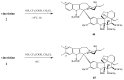
- Barnett C.J., Cullinan G.J., Gerzon K., Hoying R.C., Jones W.E., Newlon W.M., Poore G.A., Robison R.L., Sweeney M.J., Todd G.C. Structure-activity relationships of dimeric Catharanthus alkaloids. 1. Deacetylvinblastine amide (vindesine) sulfate. J. Med. Chem. 1978;21:88–96. - PubMed
-
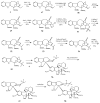
- Thimmaiah K.N., Lloyd W.D., Sethi V.S. A simple method for the chemical modification of antitumor Catharanthus Vinca alkaloids. Ind. J. Chem. Sect. B: Org. Chem. Med. Chem. 1990;29:678–680.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

